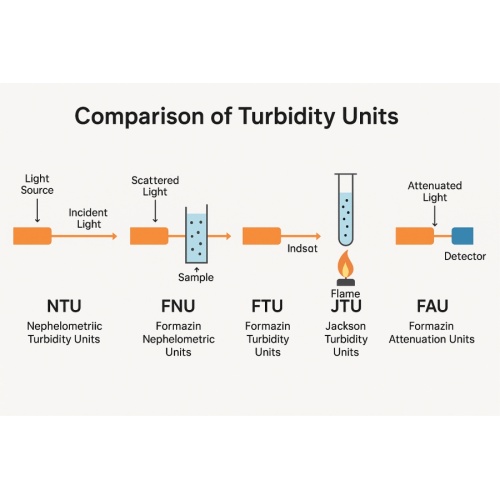
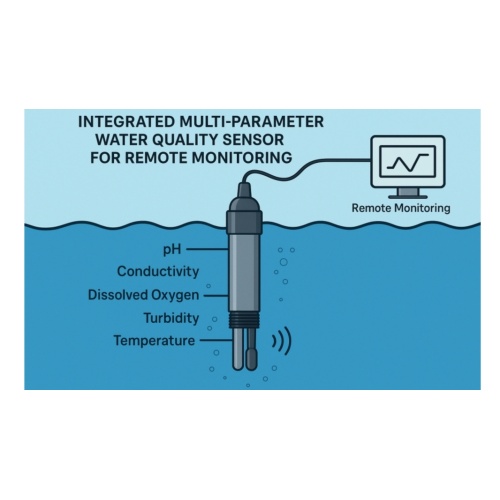
Overview
Understanding the difference between a PH Electrode and a PH Sensor is essential for accurate pH measurement in various applications, including water treatment, laboratory testing, and industrial process control. While these two terms are often used interchangeably, they refer to different components or systems within the broader scope of pH monitoring. This article will explore what a pH electrode and a pH sensor are, how they function, their differences, and where each is typically used.?

?
What is a PH Electrode?
A PH Electrode is a component of a pH measurement system that directly measures the hydrogen ion concentration in a solution. It typically consists of a glass membrane sensitive to hydrogen ions and an internal reference electrode. The electrode generates a voltage that varies depending on the pH level of the solution, and this voltage is then interpreted by a pH meter or transmitter. PH electrodes are known for their high accuracy and are widely used in laboratory and industrial settings.?
?
What is a PH Sensor?
A PH Sensor is a complete pH monitoring device that often includes a pH electrode, a reference system, a temperature sensor, and electronic circuitry for signal processing. PH sensors convert the chemical measurement of hydrogen ion activity into an electrical signal that can be transmitted to controllers or monitoring systems. They are designed for field applications and are typically rugged and ready for real-time integration in processes like water quality monitoring or wastewater treatment.?
?
Key Differences Between PH Electrode and PH Sensor
- Component vs. System: A pH electrode is a key component of a pH sensor system, whereas the sensor includes multiple integrated components such as temperature compensation and signal processing.
- Output: A pH electrode outputs a raw millivolt signal, while a pH sensor provides a processed and calibrated signal, often through analog (4-20mA) or digital (RS485/Modbus) communication.
- Application: PH electrodes are mainly used in controlled environments like laboratories, whereas PH sensors are more suitable for harsh field environments and continuous monitoring systems.
- Maintenance: Electrodes usually require more frequent calibration and maintenance, while sensors are built for long-term deployment with minimal servicing.
Applications and Use Cases
PH Electrode and PH Sensor technologies are applied across numerous industries. In laboratory research, the high precision of standalone pH electrodes is preferred. Meanwhile, PH Sensors are ideal for applications like industrial pH control, environmental water quality monitoring, aquaculture, and food processing. These pH measurement devices enable real-time, accurate readings necessary for maintaining safety and compliance in many operations.?
?
Conclusion
In summary, both PH Electrodes and PH Sensors play vital roles in pH measurement systems. While the pH electrode serves as a sensing element, the pH sensor provides a complete solution for pH monitoring. Choosing the right device depends on the application, environmental conditions, and measurement requirements. Understanding the difference between PH Electrode and PH Sensor ensures better selection, performance, and maintenance of your pH monitoring devices.?