Overview
In modern scientific research, production and environmental monitoring, the acidity and alkalinity of the solution (i.e. pH value) is a crucial indicator. In order to accurately measure and monitor this parameter, pH analyzers (also known as pH meters) came into being. This article will introduce in detail the working principle of pH analyzers and their applications in various fields.
?
Working principle of pH analyzer
The working principle of pH analyzer is mainly based on electrochemical principles, and reflects its pH value by measuring the activity of hydrogen ions (H?) in the solution. This process mainly relies on two core components: PH Sensor or pH electrode and pH controller. ?
Composition and function of pH electrode
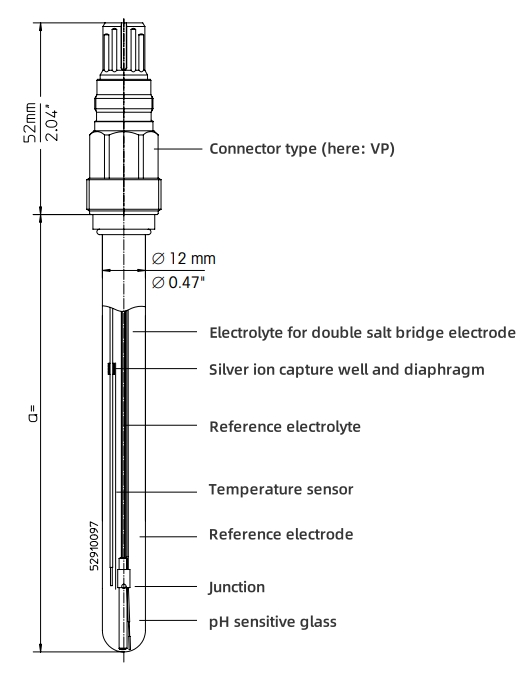
The pH electrode is the core sensor of the pH analyzer, usually made of a glass membrane that is sensitive to hydrogen ions. This glass membrane can generate a potential difference that is directly related to the pH value of the solution. When the electrode contacts the solution to be tested, the H? ions in the solution will pass through the glass membrane and form a certain concentration difference inside and outside the membrane, thereby generating a potential difference. This potential difference is linearly related to the logarithm of the H? concentration in the solution, that is, it follows the Nernst equation. By measuring this potential difference, we can indirectly know the pH value of the solution.
?
The role of pH controller
The pH controller is the "brain" of the pH analyzer, responsible for receiving, processing and displaying electrical signals from the pH electrode. First, the amplifier inside the controller converts the tiny potential difference into an electrical signal that is easy to process and transmit. Then, after a series of algorithm processing, this electrical signal is converted into an intuitive pH value and displayed on the screen of the instrument. Users can understand the pH value of the solution by reading this value. In addition, some advanced pH controllers also have functions such as automatic calibration and alarm to ensure the accuracy and reliability of the measurement.
?
Application fields of pH analyzers
Due to their high precision and convenience, pH analyzers have been widely used in many fields.
?
Laboratory application
In scientific research fields such as chemistry and biology, pH analyzers are indispensable experimental tools. It can accurately measure the pH value of various solutions and provide accurate data support for scientific researchers. This helps them to gain an in-depth understanding of important information such as the progress of chemical reactions and the metabolic activities of organisms.
?
Industrial production
In the industrial production processes of food, pharmaceuticals, chemicals, etc., the pH value of the solution is crucial to product quality. Therefore, pH analyzers are widely used in the production lines of these industries to monitor changes in pH during the production process. This helps ensure that products meet quality standards and improve production efficiency.
?
Environmental monitoring
Environmental protection is one of the important issues in today's society. pH analyzers play an important role in water quality monitoring. By measuring the pH value of water bodies, we can assess the health of water bodies, detect potential pollution problems in a timely manner, and take corresponding treatment measures. This is of great significance for protecting water resources and maintaining ecological balance.
In summary
As a high-precision, convenient and reliable pH measurement tool, pH analyzers play an important role in many fields such as scientific research, production and environmental monitoring. With the continuous advancement of technology and the continuous expansion of application fields, pH analyzers will continue to contribute to the development of human society.
?